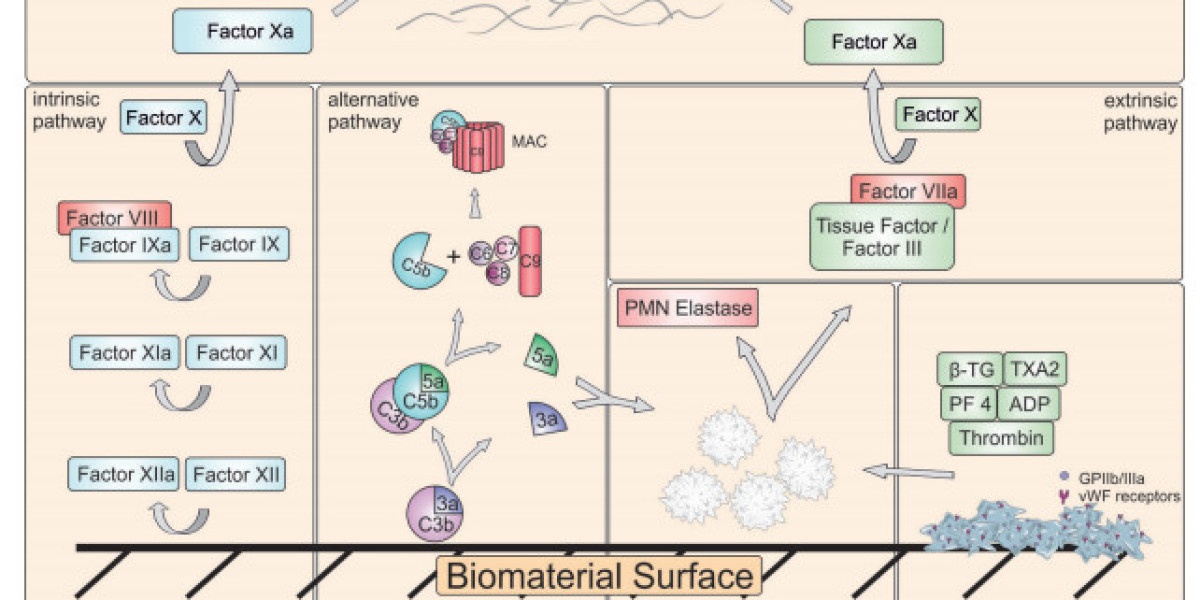
Hemocompatibility Testing | #hemocompatibility
Drug-eluting stents are typically used in cardiovascular procedures to promote the repair of diseased arteries by eluting or releasing drug products over time. Drug-eluting stents are composed of a medical device and a pharmaceutical product, so it presents unique challenges to development and regulatory approval. In addition, because they are implantable medical devices, they require additional review due to their extended contact with the human body.
STEMart provides a comprehensive suite of testing services to help manufacturer assess compliance with standards and regulatory requirements. https://www.ste-mart.com/drug-....eluting-stent-testin
The shelf life or the expiration date of a combination product is determined by the shortest estimated shelf life of the stability testing including drug stability, device aging, and packaging system/ sterile barrier aging. The stability testing provides regulatory agencies with critical information for approval, assure product quality throughout the storage period, provides health care providers with product label instructions for storage period and condition, and provides patients with confidence in the product safety.
STEMart provides a variety of stability testing services to help manufacturer determine the shelf life of their combination product. https://www.ste-mart.com/stability-testing.htm