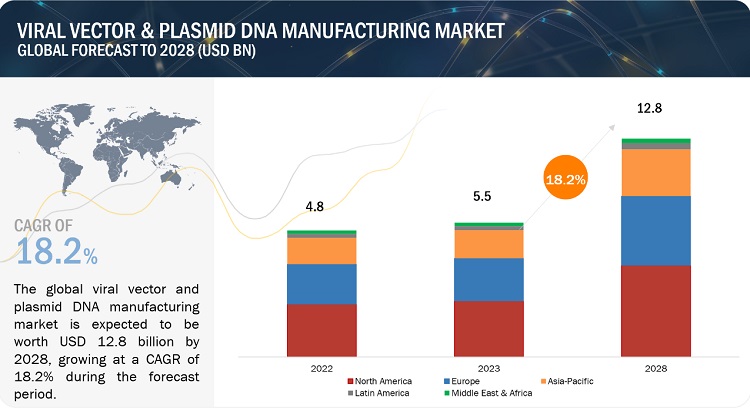
The Global Viral Vector Manufacturing Market in terms of revenue was estimated to be worth $5.5 billion in 2023 and is poised to reach $12.8 billion by 2028, growing at a CAGR of 18.2% from 2023 to 2028. The new research study consists of an industry trend analysis of the market. The new research study consists of industry trends, pricing analysis, patent analysis, conference and webinar materials, key stakeholders, and buying behaviour in the market. The growth of this market is majorly driven by g rising prevalence of target diseases and disorders, the availability of funding for gene therapy development, and effectiveness of viral vectors in gene therapy delivery. On the other hand, high operational cost associated with cell & gene therapy manufacturing is restraining the growth of this market.
Download PDF Brochure:
https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=89341986
Viral Vector Manufacturing Industry Dynamics
Driver: Effectiveness of viral vectors
Viral vectors have been proven to be effective tools for delivering therapeutic genes to target cells in gene therapy. The effectiveness of viral vectors can depend on several factors, including the type of virus used, the target cell type, the dosage and mode of delivery, and the immune response of the patient. Currently, viral vectors are the most effective gene delivery methods, especially for in vivo gene transfers. Viruses have a natural tropism to some cell types and are hence utilized in therapeutic approaches. Viral vectors can be modified according to the required application and thus prove advantageous for targeting and entering cells.
Restraint: High operational costs associated with cell & gene therapy manufacturing
Currently, more than 1,000 cell & gene therapies are in trial worldwide. There are more than 700 investigational cell & gene therapies in clinical development in the US alone. However, manufacturing facilities have not kept up. It has been estimated that hundreds of facilities will be needed to manufacture the treatments that are now in clinical trials. One of the areas that need to be accelerated is viral capacity. Most viral vectors are produced using adherent manufacturing, which is expensive to operate—a vial of 20 million cells can cost USD 20,000 to USD 30,000 to make. The cost of manufacturing for a gene therapy can be between USD 500,000 and USD 1 million, excluding the costs for R&D, the costs to run crucial clinical trials, or the costs to build the commercial infrastructure necessary to provide access to patients.
Opportunity: Leveraging digital to facilitate operational excellence
To further address operational challenges in production, manufacturers could explore the use of digital tools. One such tool is advanced analytics-based models, which can help optimize yield by proactively identifying and correcting potential yield issues. By using an analytics-driven recommendation engine built on historic deviation and root-cause data, manufacturers can shorten timelines for resolving deviations.
Challenge: Risk of mutagenesis and other unwanted outcomes
Several safety concerns have been highlighted in viral vectors that are used for cell and gene therapy manufacturing. These include inflammation, random insertion disrupting normal genes, the activation of proto-oncogenes, and insertional mutagenesis. As several factors are associated with the risk of viral vector-mediated insertional mutagenesis (viral and non-viral), a one-size-fits-all approach for reducing genotoxicity cannot be applied. Current genotoxicity testing strategies rely largely on detecting the effects on DNA (damage or mutation) following a short exposure and expression period for mutation. However, these are of limited use when considering vector-mediated insertional mutagenesis, as it takes weeks, months, or even years to manifest in patients.
The Plasmid DNA segment accounted for the largest share, by type in the viral vector & plasmid DNA manufacturing industry in 2022.
By type, the viral vector & plasmid DNA manufacturing market has been further categorized as viral vector and plasmid DNA. The plasmid DNA segment held the largest share of the global viral vector and plasmid DNA manufacturing market in 2022. This can be attributed to Increasing R&D and the growing launches of plasmid DNA production platforms. Plasmid DNA plays a crucial role in this research and developments. Plasmids are often used in genetic engineering and biotechnology for various purposes, including gene cloning, gene expression, and recombinant protein production.
The downstream manufacturing segment accounted for the largest share of the workflow segment in the viral vector and plasmid DNA manufacturing market in 2022.
Based on workflow, the global viral vector and plasmid DNA manufacturing market has been segmented into upstream manufacturing and downstream manufacturing. The downstream manufacturing segment held the largest market share in 2022. The large share of this segment can be attributed to development of efficient purification techniques and processes that result in high yields of purified products.
Asia Pacific region of viral vector and plasmid DNA manufacturing industry is estimated to register the highest CAGR during the forecast period.
Asia Pacific offers lucrative growth potential for the market. This can be attributed to the increasing pharmaceutical R&D spending, the growing trend of outsourcing drug discovery services, growing life sciences research, and increasing government initiatives for healthcare research.
One significant growth driver for the viral vector and plasmid DNA manufacturing market in the Asia Pacific region is the increasing investment in healthcare infrastructure and research & development (R&D) activities. Governments and private entities are allocating substantial funds and launching various research programs to enhance healthcare facilities and promote scientific advancements, including viral vector and plasmid DNA manufacturing technologies. These investments are driving the adoption of viral vector and plasmid DNA manufacturing techniques across various applications and research fields.
Key Market Players:
Key players in the viral vector and plasmid DNA manufacturing market include Lonza Group AG (Switzerland), Merck KGaA (Germany), Thermo Fisher Scientific Inc. (US), Charles River Laboratories International, Inc. (US), Catalent Inc. (US), WuXi AppTec (China), FUJIFILM Corporation (Japan), GenScript Biotech Corporation (US), Takara Bio Inc. (Japan), Oxford Biomedica (UK), Novartis AG (Switzerland), Precision Biosciences (US), Bluebird Bio, Inc. (US), Sartorius AG (Germany), Danaher Corporation (US), SIRON Biotech (Germany), VGXI, Inc. (US), Waisman Biomanufacturing (US), Kaneka Eurogentec S.A. (Belgium), PlasmidFactory GmbH (Germany), ATUM (US), Addgene (US), Cell and Gene Therapy Catapult (UK), Batavia biosciences (Netherlands), and Altogen Biosystems (US).
Get 10% Free Customization on this Report:
https://www.marketsandmarkets.com/requestCustomizationNew.asp?id=89341986
Recent Developments:
- In August 2022, MERCK KGaA the VirusExpress 293 Adeno-Associated Virus (AAV) Production Platform, which offers a full viral vector manufacturing offering including AAV, Lentiviral vectors.
- In May 2022, Catalent Inc., launched UpTempo Virtuoso platform process for the development and manufacturing of adeno-associated viral (AAV) vectors.
Viral Vector Manufacturing Market Advantages:
- Scalability: The viral vector manufacturing market offers scalable production processes to meet the increasing demand for viral vectors. This scalability allows for efficient and cost-effective production of viral vectors, accommodating the needs of both research and clinical applications.
- Advanced Technology: The industry leverages advanced technologies, such as bioreactor systems, single-use systems, and automation, to optimize manufacturing processes. These technologies improve productivity, enhance quality control, and streamline production, ensuring consistent and reliable viral vector manufacturing.
- Therapeutic Potential: Viral vectors serve as critical tools in gene therapy, enabling the delivery of therapeutic genes to target cells. The viral vector manufacturing market plays a pivotal role in enabling the development and production of gene therapies, offering significant therapeutic potential for a wide range of genetic disorders and diseases.
- Diverse Applications: Viral vectors find applications beyond gene therapy, including vaccine development, regenerative medicine, and cancer immunotherapy. The viral vector manufacturing market caters to these diverse applications, providing the necessary infrastructure and expertise to support research and development efforts in various fields.
- Regulatory Support: Regulatory agencies are actively involved in establishing guidelines and standards for viral vector manufacturing. This support ensures compliance with safety and quality standards, facilitating the translation of viral vector-based therapies from preclinical research to clinical applications.
- Collaborations and Partnerships: The viral vector manufacturing market encourages collaborations and partnerships among academic institutions, biotechnology companies, and contract manufacturing organizations (CMOs). These collaborations foster knowledge exchange, technology transfer, and the sharing of manufacturing expertise, driving advancements in viral vector manufacturing.
- Market Growth and Investment Opportunities: The growing demand for viral vectors in gene therapy and vaccine development fuels market growth and presents investment opportunities. The viral vector manufacturing market offers attractive prospects for investors and stakeholders, with the potential for significant returns on investment.
- Patient Impact: The viral vector manufacturing market plays a crucial role in translating scientific advancements into tangible patient benefits. By enabling the production of viral vectors, the market contributes to the development of innovative gene therapies and vaccines that have the potential to revolutionize healthcare and improve patient outcomes.
- Market Competitiveness: The viral vector manufacturing market is characterized by healthy competition among industry players. This fosters innovation, encourages continuous improvement, and drives the development of more efficient and cost-effective manufacturing processes.
- Global Reach: The viral vector manufacturing market has a global reach, serving the needs of researchers, clinicians, and patients worldwide. The availability of viral vectors on a global scale ensures that groundbreaking therapies and vaccines can reach populations in need, contributing to the advancement of global healthcare.
Overall, the viral vector manufacturing market offers numerous advantages, including scalability, advanced technology, therapeutic potential, diverse applications, regulatory support, collaborations, investment opportunities, patient impact, market competitiveness, and global reach. These factors contribute to the growth and success of the viral vector manufacturing industry, fostering the development of transformative therapies and driving innovation in healthcare.
Report Link: (Viral Vector Manufacturing Market)
About MarketsandMarkets™:
MarketsandMarkets™ is a blue ocean alternative in growth consulting and program management, leveraging a man-machine offering to drive supernormal growth for progressive organizations in the B2B space. We have the widest lens on emerging technologies, making us proficient in co-creating supernormal growth for clients.
The B2B economy is witnessing the emergence of $25 trillion of new revenue streams that are substituting existing revenue streams in this decade alone. We work with clients on growth programs, helping them monetize this $25 trillion opportunity through our service lines - TAM Expansion, Go-to-Market (GTM) Strategy to Execution, Market Share Gain, Account Enablement, and Thought Leadership Marketing.
Built on the 'GIVE Growth' principle, we work with several Forbes Global 2000 B2B companies - helping them stay relevant in a disruptive ecosystem. Our insights and strategies are molded by our industry experts, cutting-edge AI-powered Market Intelligence Cloud, and years of research. The KnowledgeStore™ (our Market Intelligence Cloud) integrates our research, facilitates an analysis of interconnections through a set of applications, helping clients look at the entire ecosystem and understand the revenue shifts happening in their industry.
Contact:
Mr. Aashish Mehra
MarketsandMarkets™ INC.
630 Dundee Road
Suite 430
Northbrook, IL 60062
USA: +1-888-600-6441
Email: [email protected]
Research Insight: https://www.marketsandmarkets.com/ResearchInsight/viral-vector-manufacturing-market.asp
Visit Our Website: https://www.marketsandmarkets.com/
Content Source: https://www.marketsandmarkets.com/PressReleases/viral-vector-manufacturing.asp



